Shopping cart
Your cart empty!
Terms of use dolor sit amet consectetur, adipisicing elit. Recusandae provident ullam aperiam quo ad non corrupti sit vel quam repellat ipsa quod sed, repellendus adipisci, ducimus ea modi odio assumenda.
Lorem ipsum dolor sit amet consectetur adipisicing elit. Sequi, cum esse possimus officiis amet ea voluptatibus libero! Dolorum assumenda esse, deserunt ipsum ad iusto! Praesentium error nobis tenetur at, quis nostrum facere excepturi architecto totam.
Lorem ipsum dolor sit amet consectetur adipisicing elit. Inventore, soluta alias eaque modi ipsum sint iusto fugiat vero velit rerum.
Sequi, cum esse possimus officiis amet ea voluptatibus libero! Dolorum assumenda esse, deserunt ipsum ad iusto! Praesentium error nobis tenetur at, quis nostrum facere excepturi architecto totam.
Lorem ipsum dolor sit amet consectetur adipisicing elit. Inventore, soluta alias eaque modi ipsum sint iusto fugiat vero velit rerum.
Dolor sit amet consectetur adipisicing elit. Sequi, cum esse possimus officiis amet ea voluptatibus libero! Dolorum assumenda esse, deserunt ipsum ad iusto! Praesentium error nobis tenetur at, quis nostrum facere excepturi architecto totam.
Lorem ipsum dolor sit amet consectetur adipisicing elit. Inventore, soluta alias eaque modi ipsum sint iusto fugiat vero velit rerum.
Sit amet consectetur adipisicing elit. Sequi, cum esse possimus officiis amet ea voluptatibus libero! Dolorum assumenda esse, deserunt ipsum ad iusto! Praesentium error nobis tenetur at, quis nostrum facere excepturi architecto totam.
Lorem ipsum dolor sit amet consectetur adipisicing elit. Inventore, soluta alias eaque modi ipsum sint iusto fugiat vero velit rerum.
Do you agree to our terms? Sign up
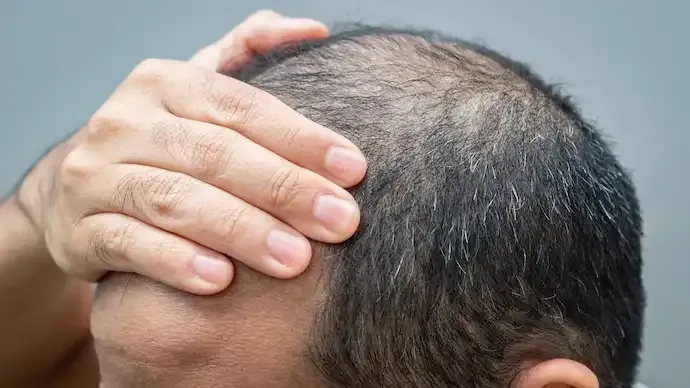
Shares of Swiss-listed Cosmo Pharmaceuticals surged nearly 40% over the past week after the company reported strong late-stage clinical results for an experimental treatment targeting male pattern baldness, sharply boosting investor confidence in its long-term growth prospects.
The rally followed the release of topline data from two large Phase III trials of clascoterone, a topical androgen receptor inhibitor being developed to treat androgenetic alopecia, the most common form of male baldness. The data marks a major milestone for Cosmo and has repositioned the company as a potential disruptor in a global hair-loss market that has seen little innovation for decades.
Cosmo said the Phase III programme demonstrated statistically significant improvements in hair growth compared with placebo, making it the largest late-stage clinical study ever conducted for a topical hair-loss treatment. The two trials involved nearly 1,500 patients and showed a meaningful increase in target-area hair count, with one study delivering particularly strong results.
Investors reacted swiftly, sending Cosmo’s shares sharply higher on the SIX Swiss Exchange as the data reduced uncertainty around the drug’s clinical effectiveness and commercial potential.
The results also strengthen Cosmo’s position beyond its existing acne franchise, where a lower-dose version of clascoterone is already approved and marketed in the United States.
If approved, clascoterone would become the first topical treatment to directly target the hormonal cause of male pattern baldness without significant systemic absorption. This sets it apart from existing treatments such as finasteride and minoxidil, which have been available for decades but often come with side effects that discourage long-term use.
By blocking dihydrotestosterone (DHT) locally at the hair follicle level, clascoterone aims to deliver efficacy while minimising the safety concerns associated with oral therapies — a critical factor in a lifestyle-driven market.
Cosmo’s next key milestone is the completion of 12-month safety data, expected in 2026. Subject to positive results, the company plans to seek regulatory approvals in the United States and Europe.
The company has also indicated it is open to partnering with larger pharmaceutical players for global marketing and distribution rather than pursuing commercialisation alone. With a solid cash position, Cosmo says it can afford to wait for the right strategic partner.
Cosmo estimates the addressable market for male pattern baldness in the US alone at more than $20 billion, with the global opportunity significantly larger. That scale explains the sharp re-rating of the stock following the Phase III data.
For investors, the surge reflects a familiar biotech pattern: when late-stage trial results meaningfully reduce regulatory and execution risk, valuations can change dramatically in a short period.
While regulatory approval is still some distance away, the latest data has firmly put Cosmo on the radar as a serious contender in one of the world’s largest lifestyle drug markets — and turned what began as a “good hair day” into a strong run for its shares.
129
Published: Dec 27, 2025